基于 4-噻唑烷酮的化学疗法与含 PEG 的聚合物纳米载体的水分散复合物增强的促凋亡作用
摘要
目标
研究4-噻唑烷酮衍生物与含PEG聚合物纳米载体复合物的水制剂是否增强其对大鼠神经胶质瘤C6细胞的促凋亡作用。
方法
在体外用大鼠神经胶质瘤 C6 细胞研究了 4-噻唑烷酮衍生物的抗肿瘤作用机制。评估细胞出生、细胞周期模式和膜联蛋白 V 表达,并通过 DNA 彗星分析估计 DNA 损伤。 4-噻唑烷酮衍生物与聚合物纳米载体复合的新型水性制剂用于增强对C6细胞的促凋亡作用。
结果
所研究的 4-噻唑烷酮衍生物使用凋亡机制杀死大鼠神经胶质瘤 C6 细胞,如 G1 前阶段这些细胞的 FACS 分析所证实的,膜联蛋白 V 阳性 C6 细胞的出现,以及更高级别的 DNA 彗星数量增加。研究的化合物与含 PEG 的聚合物纳米载体的复合显着增加了通过上述所有方法测量的大鼠神经胶质瘤 C6 细胞的促凋亡作用。
结论
4-噻唑烷酮衍生物与含PEG的聚合物纳米载体的络合使它们具有水溶性并增强了大鼠胶质瘤C6细胞的促凋亡作用。
介绍
化疗是许多肿瘤的主要治疗方式,化疗药物被选为最后的选择,尤其是在癌症已经扩散的情况下[1]。然而,严重的副作用降低了当前许多抗癌药物的临床疗效。因此,开发具有减少或最小副作用的替代性或协同性抗癌药物是持续且至关重要的需求[2]。
抗癌药物的低水溶性一直是阻碍其开发和临床应用的普遍而严重的问题,许多高活性和有前途的化疗药物因其低水溶性而被排除在外[3]。表面活性剂和助溶剂以高浓度使用以溶解水不溶性抗癌剂;然而,这些物质也会产生不良的副作用,从而限制了许多抗癌药物的使用[4]。
药物开发面临的一大挑战是消除或至少减少高活性药物的负面副作用,尤其是抗肿瘤药物在体内表现出普遍毒性,这严重限制了它们的使用 [5,6,7,8,9 ]。克服这个问题的一种有效方法是使用药物的多功能纳米载体,使有毒抗肿瘤药物能够在其递送到特定器官或组织中的靶细胞的部位结合 [8, 10, 11]。与其他材料相比,纳米颗粒的小尺寸、可控的特定结构、大表面积和形状为它们提供了几个优势 [5, 8, 9]。使用纳米粒子时,可以优化效率,最大限度地减少负面副作用,并改善癌症治疗 [12, 13]。因此,由于高一般毒性而较早失败的药物现在可以获得第二次临床使用机会,因为它们包含在提高了生物利用度和控释的递送系统中。这种药物载体复合物更容易穿透天然屏障,如血脑屏障或单个细胞的膜。纳米粒子的大表面积使它们能够与特定的载体生物分子形成复合物,有助于将药物输送到靶器官,从而显着减少甚至完全消除负面副作用,允许增加最小有效剂量单次给药,提高药物作用的有效性,为个体化药物治疗的发展提供新的动力。此外,纳米粒子可以封装分子,从而提高某些药物的溶解性、稳定性和吸收性 [11, 13,14,15]。药物递送系统可以提供药物在血液中的延长循环、药物在病理生理过程中积累的能力以及有效地将活性物质分子转移到细胞和细胞器中的能力。纳米颗粒必须稳定,在一定时间内保持其化学结构,同时能够生物降解[16, 17]。
为了评估新的抗癌药物,重要的是使用各种靶人类癌细胞系和体内实验肿瘤模型来测量它们的细胞毒活性和效力。 化疗常因细胞凋亡过程缺陷而失败,而细胞凋亡过程在肿瘤发生改变后或由肿瘤发生变化引起的药物诱导细胞死亡中起关键作用[18,19,20,21]。
由于许多恶性细胞可以逃避细胞凋亡,因此在设计和开发新的抗癌药物时应采用合理的方法。开发新型抗癌药物的主要目标是:(1)找到克服影响独立药物作用机制的单个癌细胞突变的方法; (2) 设计能够同时靶向独立通路的化疗方案。更好地了解癌症遗传学与治疗敏感性之间的关系是开发新的有效抗癌药物的关键问题[22]。
在之前的研究中,我们证明合成的 4-噻唑烷酮衍生物(Les-3288、Les-3833 和 Les-3882)可能使用与其他抗癌药物不同的作用机制在体外杀死大鼠 C6 神经胶质瘤和人 U251 胶质母细胞瘤细胞,相反阿霉素(Dox)。 Les-3288 没有显着影响处理细胞中的活性氧 (ROS) 水平 [23, 24]。应该强调的是,与 Dox 相比,这些有效的抗肿瘤药物在实验动物体内表现出较低的一般毒性,正如测量的它们在肿瘤细胞和动物中的毒性作用的生化参数所证明的那样 [7, 8]。因此,与这些物质以游离形式的作用相比,抗肿瘤药物与聚合物纳米载体 (PNC) 的结合和药物以稳定的水输送系统的形式应用可以减少对动物器官的毒性作用。 7, 8]。
这项工作的目的是研究 4-噻唑烷酮衍生物与含 PEG 的 PNC 复合物的水性制剂在体外和体内对 C6 系大鼠神经胶质瘤细胞的凋亡诱导,并比较使用这些衍生物的凋亡诱导以自由形式。
材料和方法
抗癌药物
杂环 4-噻唑烷酮衍生物(化合物 Les-3288 和 Les-3833,图 1)是在乌克兰丹尼洛·哈利茨基利沃夫国立医科大学的药物、有机和生物有机化学系合成的,如前所述 [25]。
<图片>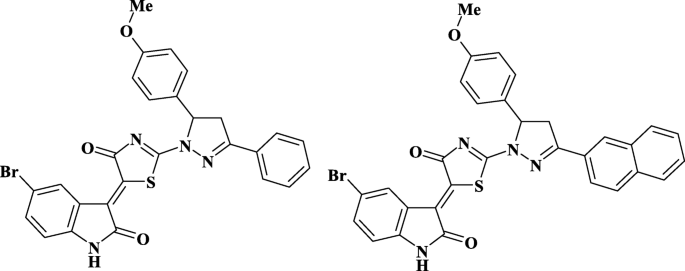
研究化合物的结构Les-3288和Les-3833
在用于细胞培养之前,将这些化合物溶解在二甲基亚砜(DMSO,Arterium,Lviv,Ukraine)中。将溶液另外在沸水浴中保持5分钟,并用蒸馏水稀释以达到工作浓度。培养基中 DMSO 的终浓度低于 0.1%。 Dox 是在当地药店从辉瑞(意大利)乌克兰代表处购买的。
聚合物纳米载体
用于给药的 PNC 是在乌克兰利沃夫国立理工大学有机化学系使用前面描述的方法合成的 [26, 27]。聚(VEP-co)的合成 -GMA)-接枝-PEG通过如下的后续阶段进行。保利(VEP-co -GMA) 由 5-tert 自由基共聚反应合成 丁基过氧-5-甲基-1-己烯-3-炔(VEP,0.41 克,0.5 摩尔)(通过所述方法合成的过氧化物单体 [28])和甲基丙烯酸缩水甘油酯(GMA,7.72 克,12.2 摩尔)(Sigma-Aldrich , USA) 在乙酸乙酯 (7.9 mL) (Merck, Darmstadt, Germany) 中使用偶氮异丁腈 (AIBN, 0.129 g, 0.05 mol) (Merck, Darmstadt, Germany) 作为自由基引发剂。聚合在 343K 下进行,直到达到 65% 的最大转化率。保利(VEP-co -GMA) 用作通过聚(乙二醇)甲基醚 (mPEG) 与侧环氧基团的加成反应连接侧 PEG 链的主链。将三氟化硼醚合物(0.027 mL,0.031 mol)(Sigma-Aldrich,St. Louis,MO,USA)在 313 K 和搅拌3小时。将 mPEG (2.5 mg, 3.35 mmol) (Sigma-Aldrich, USA) 溶解在二恶烷 (15 mL) 中,将该溶液与主链聚合物溶液混合并在 313 K 下搅拌 6 小时。使用透析袋去除未反应的 mPEG具有 6-8 kDa 的截留分子量 (MWCO) 孔径 (Sigma-Aldrich, USA)。产生的梳状聚(VEP-co -GMA)-接枝-PEG在室温下真空干燥直至恒重。 PNC 的结构和一些特性如图 2 所示,之前已经描述过 [29]。
<图片>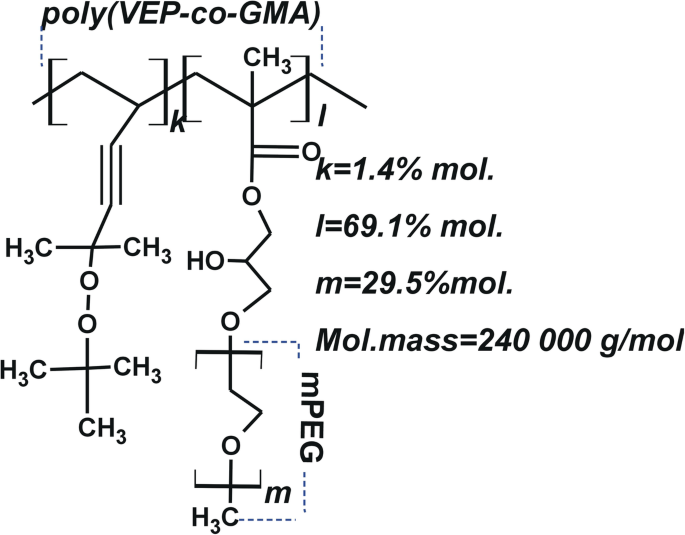
PNC 的结构示意图——poly(VEP-co- GMA)-嫁接 -PEG,主链位于 2-叔 不饱和过氧化物共聚物的基础上 -丁基过氧-2-甲基-5-己烯-3-in(VEP,用“k”表示),带有甲基丙烯酸缩水甘油酯(GMA,用“l”表示)和聚乙二醇(PEG,用“m”表示)的侧链)
为了制备水基 PNC 溶液,将 0.09 g PNC 溶解在 0.9 mL DMSO 中。将此 PNC 溶液添加到 8.0 mL 0.9% NaCl 溶液中。然后,将溶液搅拌 0.5 小时并超声处理 10 秒。通过将 4-噻唑烷酮和 PNC 的 DMSO 溶液按以下顺序滴入水中,获得复合物的水分散体:0.045 g PNC 溶解在 0.15 mL DMSO(Sigma-Aldrich,USA)中,和 0.0015 g Les -3288 (或 Les-3833) 溶解在 0.10 mL DMSO 中。将PNC和4-噻唑烷酮溶液混合后加入4.25 mL 1% NaCl水溶液中,超声10 s。
这种表面活性 PNC 包含接枝到疏水主链的亲水 PEG 链(图 2a)。在水溶液中,两亲性 PNC 形成胶束状结构,可以溶解水不溶性化合物(例如药物),或者这些化合物可以吸附到纳米粒子的表面,从而增加它们的生物相容性。为获得基于 4-噻唑烷酮的化学治疗剂的高度稳定的水分散体而开发的技术包括纳米级药物颗粒的成核,当将 DMSO 中的稀释溶液转移到含有聚合物表面活性剂 PNC 的沉淀盐水溶液中时,会发生纳米级药物颗粒的成核,为纳米粒子表面。结果,由于聚集和沉降,复合物被稳定并且分散被阻止。此外,4-噻唑烷酮衍生物的纳米级复合物受到保护,不会与周围的生物微环境发生可能的相互作用。这种保护可以让它们在体内停留更长时间,而不会失去稳定性并在目标组织或器官中积聚,而不会引起负面副作用。因此,这种修饰增强了抗癌化合物纳米颗粒的水分散体的聚集和沉降稳定性。
使用 Zetasizer Nano ZS(Malvern Instruments GmbH,Stuttgart,Germany)和 DynaPro NanoStar(Wyatt Technology,Santa Barbara, CA, USA) 和在 25°C 下使用非侵入性反向散射 (NIBS) 技术的光子相关光谱。如上所述制备用于 DLS 测量的样品,如有必要,用 pH 6.5-7.0 的双蒸水稀释。对每个样品进行三到五次测量(每次测量由 5 个循环组成,测量之间的范围为 5 分钟)。图 3 显示了 PNC 的胶束结构和 PNC 与杂环 4-噻唑烷酮(Les-3833 和 Les-3288)复合物的纳米粒子的尺寸分布,由 DLS 分析。使用透射电子显微镜 JEM-200-(JEOL,日本)在 200 kV 的加速电压下研究 PNC 胶束以及 PNC 包覆的 4-噻唑烷酮纳米粒子的尺寸和形态 [30, 31]。如上所述制备样品,如果需要,用双蒸水稀释。通过超声波分散剂 UZDN-1A(Ukrrospribor Ltd.,Ukraine)将测试溶液喷涂到基材上来制备样品,这有助于在基材上形成均匀的涂层。沉积在铜网格上的薄无定形碳膜用作衬底。通过DLS和TEM方法研究的PNC胶束和与杂环4-噻唑烷酮(Les-3288和Les-3833)的配合物的尺寸和形态如图3所示。
<图片>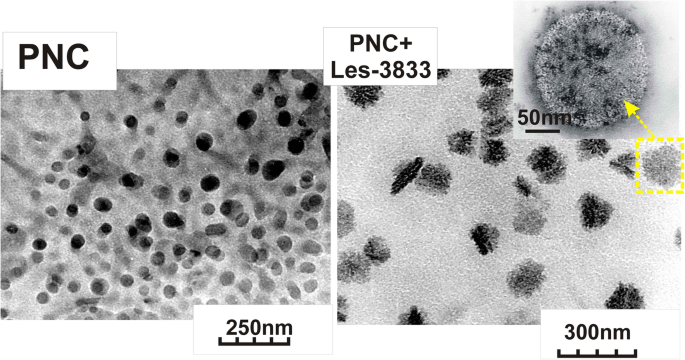
PNC(左)和PNC + Les-3833复合物(右)的透射电子显微镜(TEM)图像
DLS 和透射电子显微镜 (TEM) 研究表明 PNC 胶束的尺寸为 50 ± 25 nm,由 PNC 与杂环 4-噻唑烷酮 Les-3833 和 Les-3288 复合形成的纳米颗粒为 140 ± 25 nm 和 155 ± 30 nm,分别. PNC 与 4-噻唑烷酮衍生物的复合物的分散体高度稳定,并通过吸附在噻唑烷酮纳米颗粒表面上的 PNC 壳防止聚集和沉降。如图 4 所示,分散在水系统中的纳米颗粒在多次用水稀释以及 PNC 与 4-噻唑烷酮复合物的水系统老化 6 个月后的尺寸变化可以忽略不计。
<图片>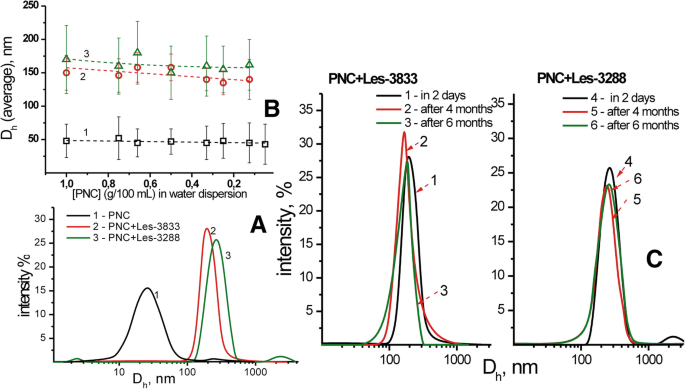
PNC 和配合物的流体动力学直径的 DLS 研究 (a ) 和 PNC-药物复合物的平均大小对分散体稀释的依赖性 (b ):PNC (1) 的水分散体、Les-3833+PNC (2) 和 Les-3288+PNC (3) 的复合物,以及 Les-3833+PNC (1-3) 和 Les- 的流体动力学尺寸3288+PNC (4–6) 分散体在储存 2 天 (1.4)、4 个月 (2.5) 和 6 个月 (3.6) (c )
细胞培养
C6 系的大鼠神经胶质瘤细胞获自乌克兰国家科学院(基辅,乌克兰)分子生物学和遗传学研究所的细胞培养物保藏中心。细胞在补充有 10% 胎牛血清(Sigma,USA)的 Dulbecco 改良 Eagle 培养基(DMEM,Sigma,美国)中培养。细胞在 CO2 培养箱中在 37°C、5% CO2 和 95% 湿度下生长。以1:5的比例每2-3天进行一次细胞补种。
研究物质的细胞毒性作用评价
将细胞以 100,000 个细胞/mL 的浓度接种在 24 孔板中。 (Greiner Bio-One North America, Inc., Monroe, NC, USA)。在细胞接种后开始的 24 小时适应期后,以 0.1、0.5 和 1.0 μM 的浓度添加所研究的物质。使用锥虫蓝染料(DV-T10282,Invitrogen,Thermo Fisher Scientific Corp.,Waltham,MA,USA)以 0.01% 终浓度,在将其加入细胞悬液后 2 分钟,在血细胞计数室中定期进行细胞计数.死细胞由于质膜受损而吸收了这种染料。
流式细胞术
为了研究细胞周期和细胞凋亡,C6 细胞用 Les-3288、Les-3833 及其与 PNC 的复合物以及游离形式的 PNC 处理,用磷酸盐缓冲盐水(PBS,pH 7.4)洗涤,离心沉淀在 4°C 下以 1000 rpm 的速度离心 5 分钟,然后重新悬浮在冷 PBS 中(每 1 mL PBS 中有 200 万个细胞)。然后,通过加入等分试样的 4 mL 冷却至 - 20°C 的无水乙醇并轻轻混合来固定细胞。固定样品保持在- 20°C直至使用(不超过1周)。对于荧光激活细胞分选 (FACS) 分析,将细胞样品在 4°C 下以 1000 rpm 的速度离心 5 分钟,弃去上清液,并将细胞沉淀物重新悬浮在 1 mL 的 PBS 中。将 100 微升不含 DNase 的 RNase(Sigma,美国)加入该悬浮液中,并将样品在 37°C 下孵育 30 分钟。然后,每个样品补充有 100 μl 碘化丙啶 (PI)(Sigma,美国,1 mg/mL),并在室温下孵育 10 分钟。最后,将样品转移到塑料 Falcon 管中,并用 FACSCalibur 流式细胞仪(BD Biosciences,Mountain View,CA,USA)和 Summit v3.1 软件(Cytomation,Inc.,Fort Collins,CO, USA)用于测量细胞周期和细胞凋亡的参数。
使用 FACS 分析进行膜联蛋白 V 阳性(凋亡)细胞的测量。大鼠神经胶质瘤 C6 细胞用 0.1 mg/mL、0.5 mg/mL 和 1 mg/mL 浓度的 PNC、Les-3288、Les-3833 及其聚合物缀合物处理,如图所示。实验结束时,用胰蛋白酶-EDTA 溶液将细胞从培养皿底部分离,用 PBS 洗涤两次,并使用细胞凋亡检测试剂盒 (BD Biosciences, San Jose, CA, USA) 用 Annexin V-FITC 染色, 根据制造商的说明。洗涤后的细胞在含有 1/50 体积 FITC 偶联的膜联蛋白 V 溶液的膜联蛋白 V 结合缓冲液(BD Pharmingen)中孵育 15 分钟。然后,将样品用适当体积的膜联蛋白 V 结合缓冲液稀释两次,并立即在流式细胞仪(Becton Dickinson,美国)的 FL1/FL2 (FITC-PI) 通道上进行测量。 Annexin V单阳性细胞为凋亡细胞。
DNA 彗星检测
DNA彗星试验在碱性条件下进行。每个样品在 75 μl 低熔点琼脂糖 (0.5%) 中混合一万个细胞。混合后,将样品移液到预先用 1.5% 正常熔点琼脂糖覆盖的载玻片上。样品在裂解溶液(2.5 M NaCl、100 mM EDTA、10 mM Tris-base、10% DMSO、1% Triton X-100)中在4°C下孵育18小时。为了使 DNA 解旋和碱不稳定损伤的表达,将载玻片在室温下在碱性溶液中孵育 20 分钟(在黑暗中进行)。对于电泳(~74 V/cm 30 分钟),将载玻片转移到水平室并添加 1x 电泳缓冲液(0.3 M NaOH、1 mM EDTA,pH> 13)。将载玻片固定在冰冷的 100% 甲醇中并干燥。载玻片用 80 μL 1×溴化乙锭 (EtBr) 染色 5 分钟,然后浸入冷却的蒸馏水中以去除多余的污渍。使用显微镜(卡尔蔡司,德国)对 DNA 彗星进行可视化,并使用软件“CASP”(Casplab-1.2.3b2 软件,CASPlab,弗罗茨瓦夫,波兰)分析图像。每个样品计数一百颗彗星。 DNA损伤根据彗尾大小分为五个遗传毒性等级:0(0-5%损伤)、1(5-25%损伤)、2(25-45%损伤)、3(45-70%损伤) ) 和 4 (超过 70% 的伤害) [32]。损伤指数(DI)的计算方法如前所述[33]。
使用甲基绿染料的 DNA 嵌入分析
使用甲基绿测定法研究了化合物 Les-3288 和 Les-3833 插入 DNA 分子的能力 [34]。三文鱼精子 DNA (10 mg/mL) 在 37 °C 下与 15 μL 甲基绿溶液(1 mg/mL 在 H2O 中)孵育 1 小时。以 1 μg/mL 的浓度加入化合物,并在 37 °C 下避光孵育 2 小时。样品的最终总体积为 1 mL。使用 BioTek 76 883 多通道显微光度计(BioTek,Winooski,VT,USA),样品在 37°C 下孵育 2 小时后,在 630 nm 处测量甲基绿的吸收。 EtBr作为阳性对照。
数据分析与统计
所有实验重复 3 次,每个变体中 3 个平行。方差分析(ANOVA)用于组间比较。 C6大鼠神经胶质瘤细胞在细胞周期不同阶段的分布数据通过单向方差分析,然后是Tukey的事后多重比较检验。所有数据均以平均值 ± SD 表示。一个 p <0.05的值被认为具有统计学意义。
结果
以自由形式和与聚合物纳米载体复合使用的 4-噻唑烷酮衍生物的细胞毒性作用(台盼蓝排除试验)
台盼蓝排斥试验的结果表明,与使用 PNC 观察到的细胞毒性相比,Les-3288 和 Les-3833 与 PNC 复合物的水基形式对神经胶质瘤 C6 细胞(处理 24 小时和 48 小时)具有增强的毒性。这些化合物的游离形式(图 5 和 6)。在 0.1 和 0.5 μM 剂量的 Les-3288 和 Les-3833 化合物与 PNC 的复合物中观察到最高的细胞毒性作用,而在这些化合物的最高剂量 (1.0 μM) 下,检测到与 PNC 的复合增强仅在 Les-3288 24 h 的情况下(图 5 和 6)。
<图片>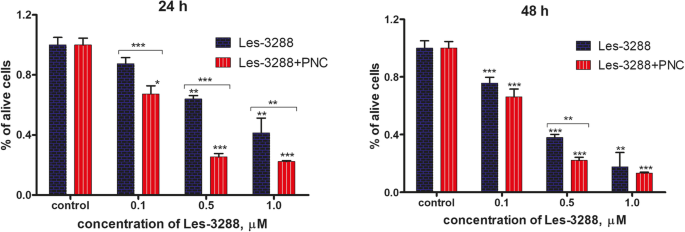
用不同浓度(0.1、0.5 和 1.0 μM)的 Les-3288 单独处理 24 小时和 48 小时后,通过台盼蓝排除试验测量的活大鼠神经胶质瘤 C6 细胞数量与其复合物与聚合物纳米载体的作用相比( PNC) *P <0.05; **P <0.01; ***P <0.001 ( 与对照相比的差异,100%)
<图片>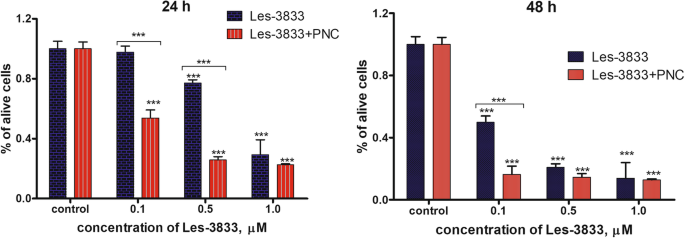
用不同浓度(0.1、0.5 和 1.0 μM)的 Les-3833 单独处理 24 小时和 48 小时后,通过台盼蓝排除试验测量的活大鼠神经胶质瘤 C6 细胞数量与其复合物与聚合物纳米载体的作用相比( PNC) *P <0.05; **P <0.01; ***P <0.001(与对照相比的差异,100%)
Les-3833 和 Les-3288 化合物与聚合物纳米载体的复合增强其体外对大鼠神经胶质瘤 C6 细胞的促凋亡活性
使用两种替代方法来评估 PNC 对所选 4-噻唑烷酮衍生物的细胞毒性潜力的调节作用。首先,进行细胞周期分析以研究药物-PNC 复合物对细胞周期的影响。此外,利用Annexin V/PI双染色来区分这些纳米复合材料诱导的细胞死亡的确切类型。
如图 7 和表 1 所示,低剂量(0.1 μg/mL 和 0.5 μg/mL)的 Les-3288 和 Les-3833 化合物对细胞周期进程或细胞凋亡诱导均无明显影响。含有 PNC 的两种化合物都显着增加了前 G1 期的细胞数量(Les-3288 的 4 倍和 Les-3833 的 6 倍),这表明它们的促凋亡潜力得到了重大提升。应该强调的是,游离形式的 PNC 对细胞周期或凋亡诱导没有影响;因此,在噻唑烷酮-纳米载体复合物处理下观察到的大量前G1细胞的出现不能用PNC和实验药物的简单协同作用来解释。
<图片>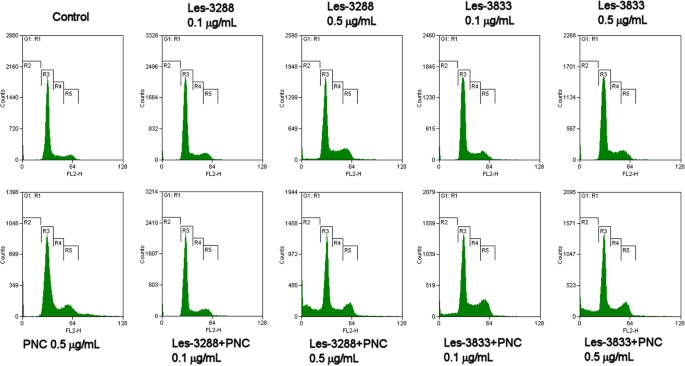
游离形式和与聚合物纳米载体 (PNC) 复合的 4-噻唑烷酮衍生物 Les-3288 和 Les-3833 在 C6 大鼠神经胶质瘤细胞中对细胞周期进程的影响。碘化丙啶 (PI) 染色,流式细胞术。 R2,前G1; R3,G1; R4,S; R5、G2阶段。 FL2-H,流式细胞仪第二通道(585/40滤光片)发射峰值
为了确认观察到的亚 G1 峰由凋亡细胞组成,分析了膜联蛋白 V/PI 染色测定的结果。在使用低剂量(0.1 和 0.5 μg/mL)和高剂量 Les-3288(1.0 μg/mL)治疗 C6 神经胶质瘤期间,我们没有观察到磷脂酰丝氨酸(早期凋亡的主要标志物,通过 FITC 偶联的膜联蛋白 V 检测)的显着外化,表明该化合物是细胞凋亡的弱诱导剂。相反,我们发现坏死细胞群增加(从对照的 1.2% 到 Les-3288 的 11.25%,1.0 μg/mL),这可能表明 Les-3288 至少部分导致坏死细胞死亡。然而,Les-3288 与 PNC 的复合完全改变了这种药物的作用方式。早期凋亡(Annexin V(+)/PI(-))和晚期凋亡细胞(Annexin V(+)/PI(+))的数量大量增加,而纯坏死细胞的总数保持在对照(未处理)细胞的水平(图 8a)。因此,将 Les-3288 与 PNC 结合不仅可以强烈增强其对神经胶质瘤细胞的促凋亡活性,而且还可以降低其潜在的促坏死细胞死亡机制。这些发现可能对临床实践具有关键意义,因为促坏死药物会大量诱导炎症,给患者带来不便,因此不推荐使用。
<图片>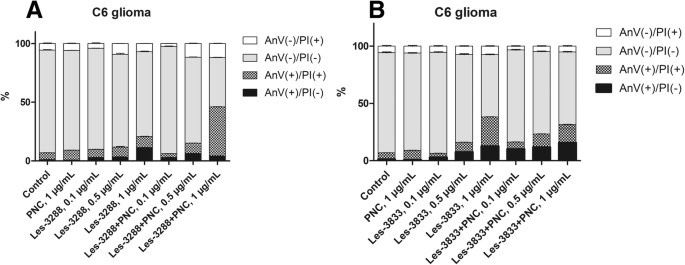
Les-3288 (a ) 和 Les-3833 (b ) 化合物及其与聚合物纳米载体 (PNC) 的复合物对大鼠神经胶质瘤 C6 细胞的影响。 Annexin V/PI 双染色,流式细胞术。 PI,碘化丙啶
令人惊讶的是,我们没有观察到 Les-3833+PNC 复合物的相同效果。事实上,在所有测试浓度下,Les-3833+PNC 复合物的促凋亡活性低于游离形式的 Les-3288(图 8b)。两种化合物之间如此巨大的差异可能是由于它们化学结构的特殊性,我们推测在Les-3833的情况下,应该优化聚合物的结构以获得更好的结果。
由自由形式并与 PNC 复合的 4-噻唑烷酮衍生物引起的大鼠神经胶质瘤 C6 细胞 DNA 损伤的彗星分析
碱性彗星试验可以检测碱性不稳定 DNA 位点的单链 DNA 断裂。使用 Olive 尾矩 (OTM) 的平均值估计获得的结果。我们的数据显示,用 Les-3288、Les-3833 和 PNC(浓度 0.5 μg/mL)处理 С6 大鼠神经胶质瘤细胞 3 小时(图 9 和 10)不会导致显着的 DNA 损伤(OTM =0.7299 ± 0.1276,OTM =1.466 ± 0.3086,OTM =0.5846 ± 0.1078),与对照中未处理的细胞相比(OTM =0.4541 ± 0.07273)。然而,用 Les-3833 + PNC 复合物 (OTM =2.3880 ± 0.2212) 进行细胞处理比用游离形式的 Les-3833 进行细胞处理造成更严重的 DNA 损伤。对于 Les-3288+PNC 复合物的作用,没有观察到这种损伤的增加(图 9 和 10)。
<图片>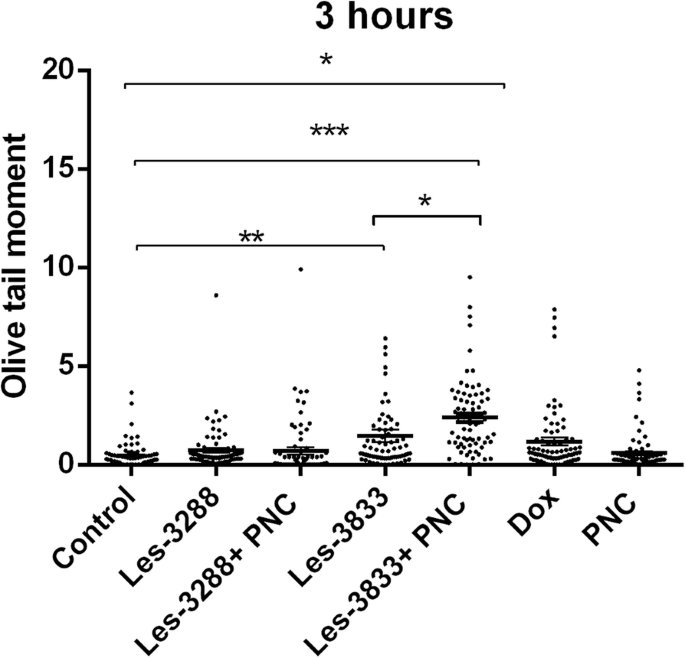
在使用浓度为 0.5 μg/mL 的实验性抗肿瘤化合物处理 3 小时的大鼠神经胶质瘤 C6 细胞中,使用 Olive 尾矩评估 DNA 损伤。 *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001。 PNC,聚合物纳米载体; Dox,阿霉素(阳性对照)
<图片>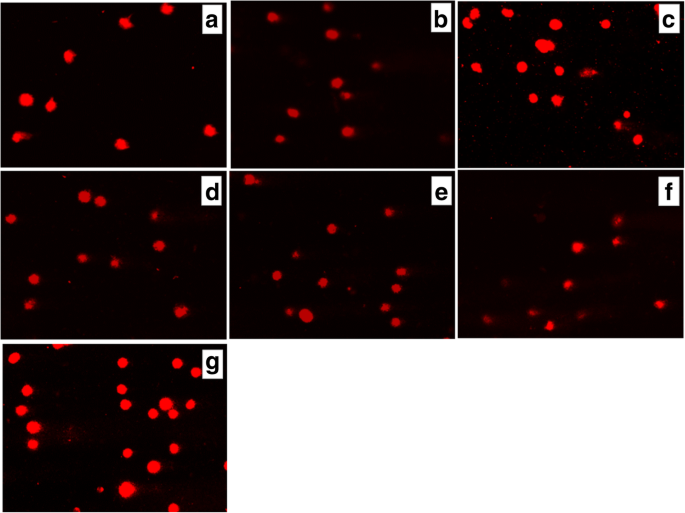
用(a处理3小时后大鼠胶质瘤С6细胞彗尾中DNA的存在 ) 对照(未经处理的细胞),Les-3288 (b ), Les-3288+PNC (c ), Les-3833 (d ) Les-3833 + PNC (e ), PNC (f ) and doxorubicin (g ), used at a 0.5 μg/mL concentration
An increase of cell incubation time to 6 h (Figs. 11 and 12) caused greater DNA damage in all experimental groups:Les-3288, Les-3288+PNC, Les-3833, Les-3833+PNC, and Dox (positive control). However, the effects of the Les-3288+PNC and Les-3833+PNC complexes appeared to be lower than the effects of the free forms of Les-3288 and Les-3833. It should be noted that the DNA-damaging effect of the complexes of both derivatives with the PNC was less than such effect from these derivatives used in free form.
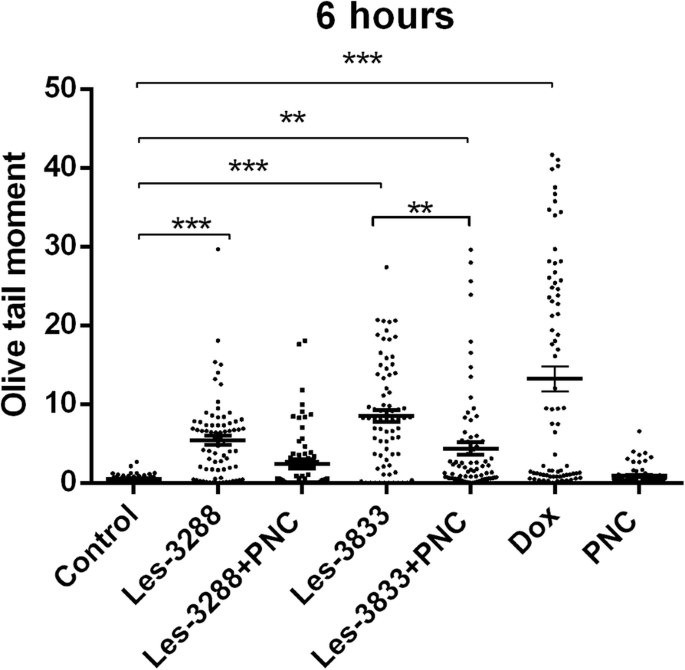
DNA damage was evaluated using the Olive tail moment in rat glioma C6 cells treated for 6 h with experimental antineoplastic compounds used at a 0.5 μg/mL concentration. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001. PNC, polymeric nanocarrier; Dox, doxorubicin (positive control)
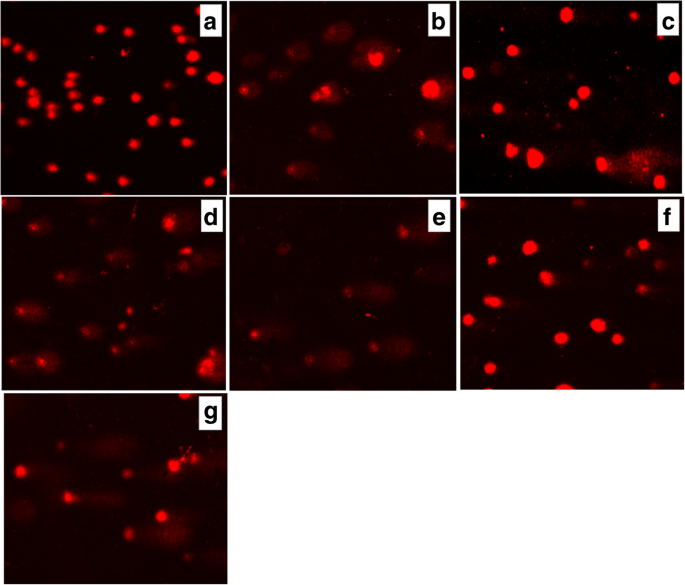
Presence of DNA in comet tail of rat glioma С6 cells after treatment for 6 h with (a ) control (untreated cells), Les-3288 (b ), Les-3288 + PNC (c ), Les-3833 (d ) Les-3833 + PNC (e ), PNC (f ), and doxorubicin (g ), used at a 0.5 μg/mL concentration
The results were evaluated using the mean value of the TailDNA%. The obtained data show that incubation of С6 rat glioblastoma cells with substance Les-3288, Les-3288+PNC, Les-3833, and PNC (all at concentration of 0.5 μg/mL) during 3 h (Fig. 13) does not lead to significant DNA damage (TailDNA% of 4.157 ± 0.682; TailDNA% of 3.530 ± 1,012, 8.807 ± 0.878; 3.298 ± 0.514, respectively) compared with control (TailDNA% =3.638 ± 0.406), but cell treatment with the Les-3833+PNC complex (TailDNA% =19.53 ± 1.48) causes more significant damage of DNA than the free form of Les-3833. An increase in the incubation time up to 6 h (Fig. 14) in all cases leads to greater damage to the DNA. Again, the level of TailDNA% detected from the action of complexes of both derivatives with the PNC was less than the level of that indicator for the action of these derivatives used in free form.
DNA damage evaluated using the % of DNA in comet tail of C6 rat glioma cells treated with experimental antineoplastic compounds during 3 h at a concentration of 0.5 μg/mL. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001. PNC, polymeric nanocarrier; Dox, doxorubicin (positive control)
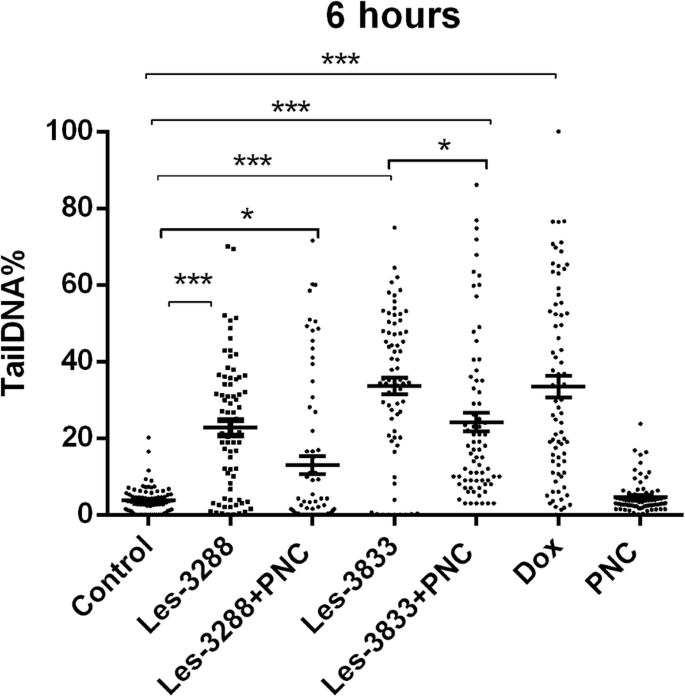
DNA damage evaluated using the % of DNA in comet tail of C6 rat glioma cells treated with experimental antineoplastic compounds during 6 h at a concentration of 0.5 μg/mL. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001. PNC, polymeric nanocarrier; Dox, doxorubicin (positive control)
We also used an alternative, five-category classification system to classify DNA migration and calculated the DI. Table 2 summarizes the percentages of rat glioma C6 cells for each level of genotoxicity during 3 h of treatment. In the control (untreated cells), higher percentages of cells in levels 0 (81.3%) and 1 (18.6%) were detected compared to these levels in Les-3288 and PNC-treated cells. Complexation of Les-3288 with the PNC did not lead to a big change in the distribution pattern of the DNA comets. In contrast, Les-3833-treated cells showed a lower percentage of cells in 0 level (38.6%) and a higher percentage of cells in level 1 (60.0%), while Les-3833 + PNC-treated cells demonstrated a significantly lower percentage of cells in level 0 (8.0%) and much higher percentages of cells with levels 1 (76.0%), 2 (13.3%), and 3 (2.6%). The integrative indicator of DNA damage (DI) in the case of application of both Les-3288 and its complex with the PNC did not differ significantly from that for control or using free PNC, while the DI from using both Les-3833 and its complex with the PNC was even higher than the DI from using Dox.
During this time interval, Les-3833 + PNC-treated cells demonstrated a higher percentage of cells with level 4 (5.3%) DNA damage than cells treated with Les-3288 and the other experimental compounds, but the percentages of cells in levels 2 and 3 were lower (16.0% and 12.0%, respectively) than those from treatment with other compounds (Table 2). Table 3 summarizes the percentages of rat glioma C6 cells with DNA damage after 6 h of treatment. In general, a tendency for the appearance of DNA comets of higher classes was detected when complexes of Les-3833 and Les-3288 were used, compared with a spectrum of DNA comet classes induced by free forms of these agents (Table 3). However, the integrative indicator of DNA damage (DI) calculated for the action of complexes of Les-3833 and Les-3288 was found to be lower than such an indicator for the action of free forms of these agents. A possible explanation could be a very high DI level at 6 h action of studied agents that is even higher than such indicator found for the action of Dox (Table 3). Thus, we observed time dependence of DNA damage caused by complexes of Les-3833 and Les-3288:at 3 h of treatment, there was an increase of the DI in the case of action of Les-3833 complexes, compared to the action of the free form of Les-3833; however, at 6 h of treatment, there was a decrease in DNA damage for the action of complexes of both Les-3833 and Les-3288, compared to such damage observed for the action of the free forms of these agents. There was no significant time dependence for the action of the PNC, while a significant time-dependent (from 3 to 6 h) increase in the DI was observed for the action of Dox (from 41.2 + 0.40 to 185.2 + 0.23).
DNA Intercalation by 4-Thiazolidinone Derivatives—Les-3288 and Les-3833
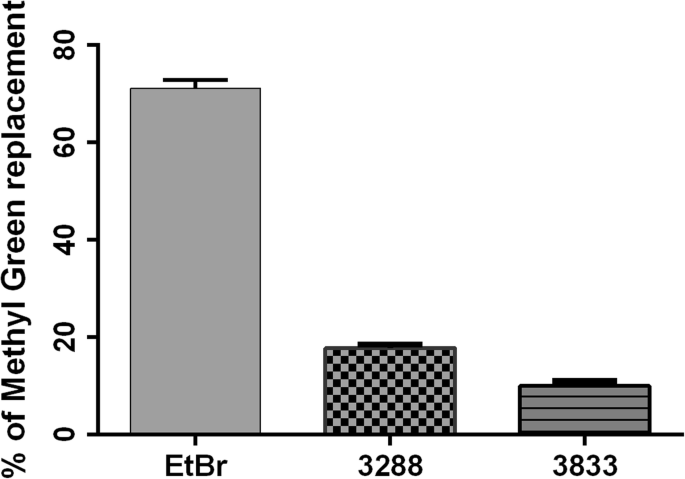
Les-3288 and Les-3833 compounds demonstrated a certain capability for intercalating DNA molecules with approximately 15% and 10%, respectively, of replacement of methyl green dye. EtBr, a well-known DNA intercalating agent, was used as a positive control, and it was shown to replace the methyl green dye much more efficiently (70%) (Fig. 15). Thus, Les-3288 and Les-3833 are not capable of effectively intercalating in between two complementary base pairs in double-stranded DNA.

Replacement of methyl green dye intercalated into the DNA of salmon sperm by Les-3288 and Les-3833 compounds (1.0 μg/mL) and ethidium bromide (EtBr) used as a positive control
讨论
There are two major problems that impede increasing the efficiency of treatment for cancer patients:(1) non-addressed actions of many anticancer drugs that lead to general toxicity and severe negative side effects due to damage of normal tissues and organs; (2) rapid (6–12 months) development of resistance of tumor cells to applied anticancer drugs that leads to a loss of efficacy of drug action. In addition, there is a third, technical problem of poor water solubility of many anticancer drugs. However, binding these drugs with specific carriers of the nanoscale size can help to solve the solubility problem.
In this study, we addressed the first and third of the above-mentioned problems by (a) selecting novel synthetic substances (4-thiazolidinone derivatives:Les-3288 and Les-3833) that possess both high anticancer activity and less general toxicity in tumor-bearing animals [7, 8, 23] and (b) applying a novel drug delivery platform that provides stable water-based forms of the complexes of the water-insoluble 4-thiazolidinone derivatives with a PEGylated polymer.
The 4-thiazolidinone derivatives—Les-3288 and Les-3833—are heterocyclic compounds with molecular mass of 400–600 Da. These and other related derivatives were synthesized at Lviv National Medical University (Ukraine), and most of them have been tested under the Developmental Therapeutic Program at the National Cancer Institute in Bethesda, MA, (USA) [35,36,37,38]. Based on these evaluations of their antineoplastic activity, the most promising substances were selected for further study of the mechanisms of their cytotoxic action and anticancer effects [25]. Les-3833 demonstrated high toxicity towards B16F10, WM793, and SK-Mel-28 melanoma cells, and against human lung A549, breast MCF-7, colon HCT116, and ovarian SKOV3 cancer cells and leukemia cells (L1210, Jurkat, HL-60 lines) [23, 25, 39]. Les-3288 and Les-3833 showed high toxicity against mammalian glioma cells (C6, U251, U373 lines) [23, 24].
Earlier, we found that Les-3288 and Les-3833 were the most toxic among the other studied 4-thiazolidinone derivatives towards rat glioma C6 cells and human glioblastoma U251 cells [23, 24], although their application for cell treatment was very complicated due to their absolute insolubility in water. Only using DMSO with additional heating was effective for preparing a soluble form of the derivatives (see the “Materials and Methods” section). Thus, it was reasonable to search for a way of improving the delivery of these derivatives to target cells, and we used a synthetic PNC to accomplish this task. The complexation of the studied derivatives of 4-thiazolidinone with the applied PNC also enhanced the effectiveness of their antineoplastic action. The mechanisms of the pro-apoptotic action of these compounds were demonstrated using western blot and FACS analyses [23, 24]. It should be noted that in healthy experimental animals, Les-3288 and Les-3833 produce much fewer negative side effects such as cardiotoxicity [40], hepatotoxicity [8], and nephrotoxicity [7] than the widely used anticancer chemotherapeutic agent Dox.
In a previous study, we showed that Les-3288 did not induce ROS production during its pro-apoptotic action in vitro and effects in experimental laboratory animals [23], while the action of Les-3833 led to such production; however, it was less than that of Dox. ROS are considered to be the main effectors of negative side effects of anticancer drugs, particularly Dox [41, 42]. Thus, the Les-3288 and Les-3833 compounds could be prospective anticancer drugs because they possess antitumor activity, but do not demonstrate general toxicity at the level characteristic for effective anticancer medicines such as Dox.
A great impediment to promoting Les-3288 and Les-3833 as anticancer drugs is their poor water solubility. This problem also exists for other anticancer medicines, such as taxols [2, 4]. For paclitaxel, this problem was solved by using a special oil for preparing the medicinal formulation [2, 4, 43]. However, specific delivery platforms are considered to be more promising for creating “smart” drugs that possess effective action in the organism [3, 13]. Specific functional polymer surfactants with block, comb-like, and block/branched architectures, including PEG-containing ones, as well as derived water forms have been developed and proposed for application as carriers for the delivery of Dox [26, 27] and the metal chemotherapeutic agent KP-1019 [10] at the Department of Organic Chemistry of Lviv Polytechnic National University.
Since the 4-thiazolidinone derivatives are water-insoluble compounds, DMSO was used to prepare their water-soluble forms. To avoid the negative consequences of using a toxic solvent like DMSO, we complexed these derivatives with the above-noted PNC. Highly dispersed and stable nanoscale water dispersions of these substances were obtained and applied for treatment of rat glioma C6 cells.
The use of complexes of 4-thiazolidinone derivatives with the PNC led to reduced general toxicity of these compounds in experimental animals. Earlier, we found that when experimental antitumor agents (Les-3288, Les-3833, Les-3882) were applied in a complex with a synthetic polymeric carrier, their action was accompanied by much smaller changes in the biochemical parameters in blood serum that are characteristic for cardiotoxicity [40], hepatotoxicity [8], and nephrotoxicity [7], compared to those for Dox. Thus, complexation of these antitumor agents with the PNC and their application in the form of water-soluble stable delivery systems reduces their toxic effects in experimental animals, compared with the action of these substances in a free form [44]. Several systems for paclitaxel delivery have been proposed including polymeric nanoparticles, lipid-based formulations, polymer conjugates, inorganic nanoparticles, carbon nanotubes, nanocrystals, and cyclodextrin nanoparticles [43].
Thus, complexation of Les-3288 with the PNC significantly increased the pro-apoptotic activity of this experimental drug, thereby allowing us to advance it to pre-clinical studies on animal tumor models. Complexation of Les-3833 with the same type of nanocarrier was less efficient, and thus another type of polymer should be used.
The developed water-based forms of the complexes, as well as the free derivatives, were applied for treatment of rat glioma C6 cells. It was found that these water-based forms of the complexes of the PNC with the 4-thiazolidinone derivatives—Les-3288 and Les-3833—enhanced the pro-apoptotic action of these compounds. In another study, we demonstrated that the induction of apoptosis by Les-3288 was not accompanied by ROS induction, opposite to the action of Dox and Les-3833 [23]. Thus, high antitumor activity of Les-3288 together with its low general toxicity when targeting the malignancy in NK/Ly lymphoma-bearing mice suggest great potential for this experimental anticancer compound. The presented data also predict the potential usefulness of Les-3288 and Les-3833 compounds complexed with the PNC for glioma treatment.
Cell division, differentiation, and death are principal physiological processes that regulate tissue homeostasis in multicellular organisms. A disruption of genomic integrity and impaired regulation of cell death may both lead to uncontrolled cell growth. A compromised cell death process can also promote genomic instability. It is becoming clear that dysregulation of the cell cycle and cell death processes plays a pivotal role in the development of major disorders such as cancer, cardiovascular disease, infection, inflammation, and neurodegenerative diseases [45].
We have found that some of the 4-thiazolidinone derivatives, namely, Les-3288 and Les-3833, were even more effective than Dox in the induction of apoptosis in rat C6 glioma and human U251 glioblastoma cells in vitro [23, 24]. It should be noted that brain tumors belong to malignancies that are the most resistant to applied chemotherapies, and survival rates in patients with these tumors are extremely low [44].
It has been shown that the complexation with this PNC enhanced the pro-apoptotic action of 4-thiazolidinone derivatives (Les-3288 and Les-3833) [44]. In another study [23], we demonstrated that the induction of apoptosis by Les-3288 was not accompanied by ROS induction, opposite to the action of Dox and partially opposite to that of Les-3833. These data could explain the lower general toxicity of Les-3288 compared with that of Dox [23, 24].
The results of FACS analysis are in agreement with the suggestion that an enhancement of the pro-apoptotic action of the studied 4-thiazolidinone derivatives is due to their complexation with a novel PNC. Such complexation of Les-3288 and Les-3833 increased the ratio of pre-G1 cells in the population of treated rat glioma C6 cells, decreased the ratio of G1 cells, and increased the action of G2/M cells. These data suggest an enhancement of both cytotoxic action (apoptosis) and cytostatic action (block in G2/M phase of cell cycle) of the studied derivatives complexed with the PNC. In general, the apoptosis mechanism of glioma cell killing by the Les-3288 + PNC complex and the Les-3833 + PNC complex was demonstrated by the results of FACS analysis of the appearance of Annexin V single-positive rat glioma C6 cells.
Involvement of the apoptosis mechanisms in the action of the studied 4-thiazolidinone derivatives was also confirmed by the results of the DNA comet assay. It was found that the complexation of Les-3833 with the PNC led to a decrease (compared to free Les-3833) in the ratio of rat glioma C6 cells in “0” DNA comets and sіmultaneous increase in the ratio of “1,” “2,” and “3” comets (3-h treatment of cells). At 6 h of treatment by the Les-3833+PNC complex, there was a large increase (compared to free Les-3833) in the ratio of “1” comets, an increase in the ratio of cells with the most expressed DNA damage (“4” comets), and a decrease in the ratios of “2” and “3” comets. It should be noted that the DNA damaging effects of Les-3288 and Les-3833 measured at 6 h was higher than such effects of complexes of these derivatives with the PNC (see Table 3). A possible explanation here could be a very high level of the DI at 6 h action of studied agents that is even higher than the level of that indicator found for the action of Dox (Table 3). There was no time dependence in the action of the PNC, while a significant time-dependent (from 3 to 6 h) increase (from 41.2 + 0.40 to 185.2 + 0.23) in the DI was detected for the action of Dox.
Thus, a complexation of 4-thiazolidinone derivatives—Les-3288 and Les-3833—with a PNC enhanced the cytotoxic effect of these compounds towards rat glioma C6 cells, and that effect was achieved through the apoptosis mechanisms including DNA damage observed as DNA comets in the electrophoresed cells.
We used the DNA intercalation test to investigate the direct targeting of DNA by the 4-thiazolidinone derivatives and revealed only weak replacement of methyl green dye (15% and 10% replacement for Les-3288 and Les-3833, respectively) that intercalated into the DNA of salmon sperm, compared to the replacement by EtBr, used as a positive control (70%).
Thus, conducting the cytotoxicity experiments in vitro with the 4-thiazolidinone derivatives—Les-3288 and Les-3833—was more convenient due to using the water-based forms of the 4-thiazolidinone derivatives—Les-3288 and Les-3833—complexed with the novel PNC. Besides, we have demonstrated that the novel synthetic 4-thiazolidinone derivatives (Les-3833 and Les-3288) possessed treatment effects i n vivo towards NK/Ly lymphoma grafted to BALB/C mice, and those effects were comparable to such effects of Dox. At the same time, the action of these derivatives led to much fewer negative side effects measured as changes in the number of erythrocytes, neutrophils, and lymphocytes in the animals’ blood and the activity of aspartate and alanine aminotransferases in their blood serum, compared with the action of Dox.
结论
The complexation of Les-3288 and Les-3833 with the PEG-containing PNC enhanced their cytotoxic action towards rat glioma C6 cells. The cytotoxic action was realized by apoptotic mechanisms and confirmed by FACS analysis as the presence of the pre-G1 fraction in the rat glioma C6 cells and of Annexin V-single-positive cells. DNA comet analysis revealed single-strand brakes in the nuclear DNA of treated glioma C6 cells that probably was not caused by the intercalation of the studied compounds into the DNA molecule. The Les-3288 and Les-3833 stabilized by the amphiphilic PEG-containing PNC resulted in the water-based forms and provided enhancement of their antitumor effect in vitro in comparison with the free form of the drugs.
缩写
- DMSO:
-
Dimethylsulfoxide
- Dox:
-
阿霉素
- EtBr:
-
Ethidium bromide
- FACS:
-
Fluorescence-activated cell sorting
- ROS:
-
活性氧
- PBS:
-
磷酸盐缓冲盐水
- PEG:
-
Polyethylene glycol
- PI:
-
碘化丙啶
- PNC:
-
Polymeric nanocarrier
- SEM:
-
扫描电镜
- TEM:
-
透射电子显微镜
- VEP:
-
2-Tret-butylperoxy-2-methyl-5-hexen-3-in
纳米材料
- 使用 Zn-Al 层状双氢氧化物光催化增强阳离子染料去除
- 基于光子晶体的混合液晶电池中增强的非线性光学效应
- 使用多功能 GaN/Fe 纳米颗粒靶向内皮细胞
- 具有增强光催化性能的新型 Bi4Ti3O12/Ag3PO4 异质结光催化剂
- Ag 纳米颗粒/BiV1-xMoxO4 与增强的光催化活性的协同效应
- 使用 Er3+-Yb3+-Li+ 三掺杂 TiO2 上转换材料提高钙钛矿太阳能电池的功率转换效率
- 使用全碳 3D 纳滤膜去除水中的抗生素
- 基于与铝/聚四氟乙烯纳米层压板集成的增强能量性能
- 金属硫化物纳米晶体在聚(3-己基噻吩)中的原位生长:[6,6]-苯基 C61-丁酸甲酯薄膜具有增强光电流的倒置混合太阳能电池
- 基于紫杉醇的靶向脂质纳米颗粒的抗增殖和细胞凋亡触发潜力,通过转铁蛋白受体增强细胞内化——白血病细胞研究
- 通过快速硒化方法增强 Sb2S3 平面异质结太阳能电池的光伏特性
- 具有增强有效光吸收的针结纳米锥阵列太阳能电池的光伏性能


